At low concentrations it causes eye and nose irritations. This gas is very dangerous, as it can cause death at very high concentrations. 
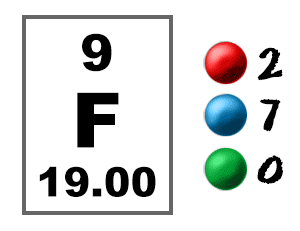
If fluorine is absorbed too frequently, it can cause teeth decay, osteoporosis and harm to kidneys, bones, nerves and muscles.įluorine gas is released in the industries. Fluorine can also protect us from dental decay, if it is applied through toothpaste twice a day. Large quantities of fluorine can be found in tea and shellfish.įluorine is essential for the maintenance of solidity of our bones. Fluorine can be found in any kind of food in relatively small quantities. As a result humans are exposed to fluorine through food and drinking water and by breathing air. Small amounts of fluorine are naturally present in water, air, plants and animals. Up to 50 ppb has been recorded in city environments. In the atmosphere 0.6 ppb of fluorine are present as salt spray and organicochloride compounds. When fluorine is attached to very small particles it can remain in the air for a long period of time. Fluorides that are found in air will eventually drop onto land or into water. Hydrogen fluorides can be released into air through combustion processes in the industry. Some solis can have as much as 1000 ppm and contaminated solis have been found with 3500 ppm. Soils contain approximatively 330 ppm of fluorine, ranging from 150 to 400 ppm. Fluorine is the 13th most aboundant element in the Earth's crust: 950 ppm are contanined in it. Fluorides are released into the air in wind-blown soil. The main mining areas for fluorite are China, Mexico and Western Europe.įluorine occurs naturally in the earth's crust where it can be found in rocks, coal and clay. Fore more information visit our page on mineral water.Īnnual world production of the mineral fluorite in around 4 million tonnes, and there are around 120 million tonnes of mineral reserves. Fluorochlorohydrocarbons are used extensively in air conditioning and in refrigeration.įluorides are often added to toothpaste and, somewhat controversially, to municipal water supplies to prevent dental cavities. Fluorides are compounds that combine fluoride with some positively charged counterpart.Ītomic fluorine and molecular fluorine are used for plasma etching in semiconductor manufacturing, flat panel display production and MEMs fabrication.įluorine is indirectly used in the production of low friction plastics such as teflon and in halons such as freon, in the production of uranium. In aqueous solution, fluorine commonly occurs as the fluoride ion F. It is so reactive that glass, metals, and even water, as well as other substances, burn with a bright flame in a jet of fluorine gas. Fluorine readily forms compounds with most other elements, even with the noble gases krypton, xenon and radon. Separation and Concentration Purification Requestįluorine - F Chemical properties of fluorine - Health effects of fluorine - Environmental effects of fluorineįluorine is an univalent poisonous gaseous halogen, it is pale yellow-green and it is the most chemically reactive and electronegative of all the elements.Plant Inspection & Process Optimalisation.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
